The western blot (also called the protein immunoblot), or western blotting, is a widely used analytical technique in molecular biology, and immunogenetics to detect specific proteins in a sample of tissue homogenate or extract. Besides detecting the proteins, this technique is also utilized to visualize, distinguish, and quantify the different proteins in a complicated protein combination. The western blot method is composed of a gel electrophoresis to separate native proteins by 3-D structure or denatured proteins by the length of the polypeptide, followed by an electrophoretic transfer onto a membrane (mostly PVDF or nitrocellulose) and an immunostaining procedure to visualize a certain protein on the blot membrane. (Source: Wikipedia https://en.wikipedia.org/wiki/Western_blot).

As the procedure is complicated, the results are also impacted by diverse factors. This article will summarize the usually encountered problems and the solution in accordance.
1. No bands or no target bands on the membrane 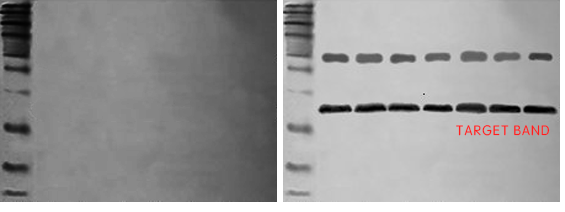
| Problem |
Possible
reasons |
Suggestion |
| Operation Problem |
Over-rinsing |
The
rinsing time should not be too long, and the detergent should not be too
strong or too much. Try to reduce the number of rinsing times, rinsing time
and the temperature. |
| The protein has not been transferred to the
membrane |
After
the transfer, determine the ransfer efficiency with the total protein stain;
during the transfer process, the gels should be in complete contact with the
membrane; the transfer “sandwich” is arranged correctly; wet the membrane
according to the instructions of the membrane manufacturer; transfer parts
not overheating during the electro-blotting process; stain the membrane with
Ponceau and combine with staining gel (Coomassie Brilliant Blue) to determine
whether the band is transferred to the membrane or over-transfer; adjust
appropriately the transfer time and current . |
| The protein has not fully been transferred to
the membrane |
Low-molecular-weight
antigens may pass through the transfer membrane. We can use a small pore size
membrane. |
| Over-blocking |
Reduce
the blocking time, try different blocking fluid. |
| Substrate incubation time is too short |
Extend
the substrate incubation time. |
| Antibodies |
Primary
antibody and secondary antibody do not match |
Choose
the type of secondary antibody which matches the primary antibody. The
secondary antibody must be of the same species as the primary antibody host.
The effectiveness of the secondary detection system can be verified by
setting internal parameters. |
| Primary antibody failure |
Use
the antibody within the validity period and store it in aliquots to avoid
repeated freezing and thawing. The working fluid should be freshly prepared
before use. |
| Antibodies combined to the target protein are
insufficient |
Reduce
the dilution of the primary antibody; use a high-effective primary antibody;
extend the antibody incubation time; fully immerse the membrane in the fluid. |
| Low concentration of antibodies |
Increase
the concentration of antibodies |
| High concentration of antibodies |
Using
too much primary or secondary antibody may cause rapid depletion of the
substrate and showing a weak signal |
| Antigen |
The
primary antibody does not recognize the target protein |
Compare
the antigen sequence and protein sequence with reference to the database,
select the appropriate primary antibody |
| Insufficient or degraded antigen |
The
sample amount in each lane should not be less than 20~30 ug, use protease
inhibitor and set a positive control. |
| Blocking substrate may contain proteolytic
enzyme activity |
| Blocking fluid |
The
blocking fluid cross-reacts with the primary or secondary antibody |
Use
appropriate blocking fluid |
| Buffer contains Sodium azide |
Sodium
azide is an inhibitor of HRP. Sodium azide cannot be used as a preservative
in buffers |
| ECL luminescent fluid is inactivated or not
sensitive enough |
Use
fresh and sensitive ECL |
| Insufficient exposure time |
Extend
exposure time |
2. Target bands emit weak signals 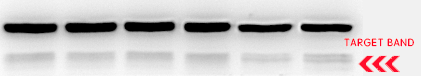
| Problem |
Possible
reasons |
Suggestion |
| Operation |
The
protein has not fully been transferred to the membrane |
Standardize
the operation of membrane transfer; optimize the conditions of
electrotransfer; two membranes should be superimposed together to prevent
excessive transfer, or use a small pore membrane. |
| Over-rinsing |
The
rinsing time should not be too long, and the detergent should not be too
strong or too much. Try to reduce the number of rinsing times, rinsing time
and the temperature. |
| Over-blocking |
Reduce
the blocking time, try different blocking fluid. |
| Insufficient exposure time |
Extend
exposure time |
| Insufficient antibody staining |
Extend
incubation time or increase antibody concentration |
| Antibodies |
Low
concentration of antibodies |
Increase
concentration of antibodies |
| Insufficient affinity between antibody and
antigen |
Increase
concentration of antibodies |
| Insufficient antibody activity |
Use
the antibody within the validity period and store it in aliquots to avoid
repeated freezing and thawing. The working fluid should be freshly prepared
before use. |
| Antigen |
Insufficient
antigen |
Increase
sample volume |
| Blocking fluid |
Buffer
contains Sodium azide |
Sodium
azide is an inhibitor of HRP. Sodium azide cannot be used as a preservative
in buffers. |
3. The background of the images is too high. 3.1 Uniform high background 
| Problem |
Possible
reasons |
Suggestion |
| Operation |
Temperature
too high during blocking,blocking is insufficient |
Selection
and optimization of the blocking fluid; use 5% skimmed milk powder; optimize
the blocking time and temperature |
| Membranes |
Prevent
membranes drying; use NC membrane for experiment; use clean tweezers and wear
gloves during operation |
| Insufficient rinsing during incubation |
Properly
increase the time and frequency of washing the membrane; ensure that the
membrane is incubated under sufficient shaking conditions and the antibody
fully covers the membrane surface |
| Overexposure |
Shorten
the exposure time, or use imaging systems |
| Antibodies |
High
concentration of antibodies |
Increase
the dilution of primary and secondary antibodies and optimize the dilution
conditions |
| The blocking fluid cross-reacts with the
primary or secondary antibody |
Replace
the blocking fluid; replace the secondary antibody or reduce the
concentration of the secondary antibody |
| Long storage time or improper storage
conditions cause antibody inactivation |
Use
fresh antibodies |
| Antigen |
Not
enough detergent in the buffer |
Increase
the concentration of Tween 20 in TBST |
| Blocking fluid |
Transfer
fluid, blocking fluid are not fresh or contaminated |
Fluid
should be freshly prepared before use. |
3.2 Heterogeneous high background 
| Problem |
Possible
reasons |
Suggestion |
| Operation |
Temperature
too high during blocking,the blocking procedure is insufficient |
Selection
and optimization of the blocking fluid;
optimize the blocking time and temperature |
| The membrane is not properly wetted and may
have dried |
Use
clean tweezers and wear gloves to operate; use enough liquid to keep the
membrane moist; use a decolorizing shaker during incubation; avoid
overlapping membranes and covering each other |
| Antibodies |
High
concentration of antibodies |
Increase
the dilution of primary and secondary antibodies and optimize the dilution
conditions |
| Secondary antibody aggregation |
Filter
the secondary antibody before use or replace it |
| Blocking fluid |
Aggregates
in the blocking fluid |
Filter
the blocking fluid before use or replace it |
| The blocking fluid cross-reacts with the
primary or secondary antibody |
Replace
the blocking fluid; replace the secondary antibody or reduce the
concentration of the secondary antibody |
| Transfer fluid, blocking fluid are not fresh or
contaminated |
Fluid
should be freshly prepared before use. |
4. A number of none-Specific bands 
| Possible
reasons |
Suggestion |
| Non-specific binding of antibodies to proteins |
Replace
antibodies |
| Concentration of Antibody is too high |
Reduce
the sample amount; increase the dilution of the primary antibody. |
| Antibody is not purified |
Use
monoclonal or affinity chromatography purified antibodies to reduce
non-specific bands |
| Strong transfer intensity of membrane causes strong signal of non-specific bands |
Reduce
the time and current of transfer |
| Blocking is incomplete |
Increase
protein concentration in blocking fluid |
| Too many passages of cells lead to differentiation of their
protein expression patterns |
Use
the original cell line or the cell line with few passages as a reference with
the current cell line |
5. Shape of the bands is abnormal 5.1 Dumbbell shape 
a. The gel is not uniform after solidification. We can recast the gel and ensure the quality of it. b. The sample may contain too many impurities. We can centrifuge the sample before use to remove the excessive impurities.
5.2 Bands are white 
The possible reason for this phenomenon is that the concentration of the first antibody is too high, the catalytic activity of HRP on the second antibody is too strong, and the chromogenic substrate is at the critical point, the reaction time is not long, and the surrounding substrate is catalysed completely. Therefore, the concentration of the first and second antibodies can be reduced, or the substrate can be changed.
5.3 Bands connect with each other 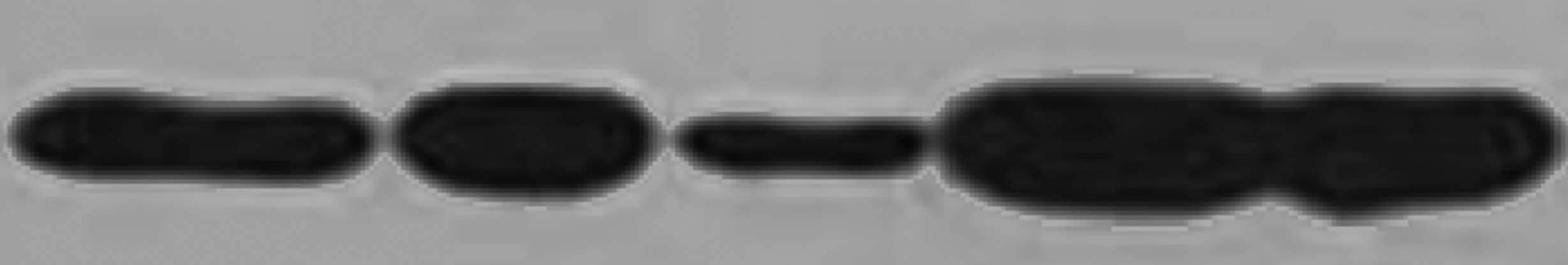
The possible reason for this phenomenon is that the amount of the sample loading is too high or there is a problem by gel casting. As a result, there occurs a gap between the separating and concentrating gels, or the sample is porous. This problem can be avoided by reducing the amount of sample loading or improving the quality of the gel casting.
5.4 Bands have tails 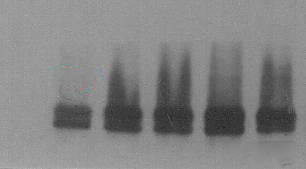
The main reason for this phenomenon is that the concentration of the protein or the first antibody is too high, or the Incubation time of the first antibody is too long.
5.5 Band is smiling 
The electrophoresis speed is too fast, and it can be slowed down by reducing the voltage. It is also possible, that the temperature of the electrophoresis is too high, which makes the gel deformed. Electrophoresis can be carried out in a cold room or ice bath.
5.6 A white circel in your band 
This phenomenon is probably due to the existence of bubbles between the membranes and the gels during the tranfering.
At last, we hope, all the researchers will bring us nice images of western blot.
Shenhua Science Technology Co.,Ltd. www.shenhuabio.net |